 
Resonance has a significant impact on the stability, reactivity, and bonding of molecules and ions, allowing for a more comprehensive understanding of their properties. It involves the existence of multiple resonance structures and the average of their contributions to the actual electronic structure. 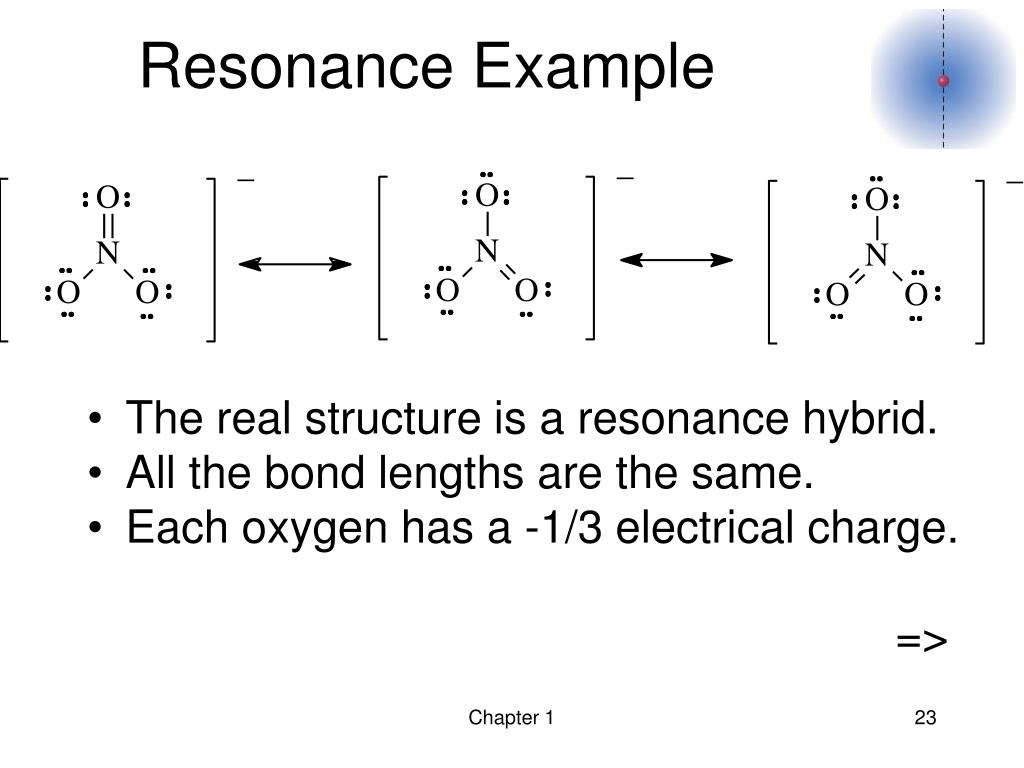
In summary, resonance is a concept used in chemistry to describe the delocalization of electrons within a molecule or ion. For example, the delocalization of electrons in the phenoxide ion (the conjugate base of phenol) stabilizes the negative charge on oxygen, making it less reactive towards electrophiles compared to a simple alkoxide ion. It can influence the stability of intermediates in chemical reactions and affect the ease with which certain reactions occur. Resonance also plays a crucial role in determining the reactivity of molecules. This is exemplified in the case of the nitrate ion (NO3^-), where the nitrogen-oxygen bonds are found to have bond orders between single and double bonds. In molecules with resonance, the actual bond order between atoms may be fractional, indicating that the electrons are shared between them. They show the different ways in which the electrons can be distributed, allowing for a more accurate representation of the molecule's electronic structure. Resonance structures are used to describe the bonding in molecules or ions that cannot be described by a single Lewis structure. The delocalization of electrons in benzene gives it exceptional stability and unique properties. In the case of the benzene molecule, for example, the electrons in the pi bonds are delocalized over all six carbon atoms. 
This delocalization leads to increased stability and lower energy for the molecule or ion. Resonance involves the delocalization of electrons, meaning that the electrons are not confined to a specific bond or atom but are spread out over multiple atoms. The actual structure of the carbonate ion is an average of these resonance structures, and the carbon-oxygen bonds are found to be of equal length. For example, in the case of the carbonate ion (CO3^2-), there are three resonance structures that can be drawn, each with a double bond between one oxygen and the carbon atom. During Paulings series of articles on the Nature of the chemical bond (NCB), his exposition of the term resonance emphasized the reality of the hybrid. They can be visualized as different ways of distributing electrons within a molecule or ion without breaking any covalent bonds. Resonance structures are different representations of a molecule or ion that differ only in the placement of electrons. Resonance occurs when there are multiple valid Lewis structures that can be drawn for a molecule or ion, and the actual structure is an average of these resonance structures. It is used to explain the electronic structure and reactivity of certain compounds that cannot be adequately described by a single Lewis structure. Resonance is a fundamental concept in chemistry that describes the delocalization of electrons within a molecule or ion. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
